The following Guide will help you understand the importance of the proper documentation and completion of the eight critical process gates for high-quality medical PCBs over and above the mere total of process steps completed on the PCB (raw count). Each gate is mapped to its relevant failure mechanism, validation requirement, procurement evidence, and compliance expectation at the 2026 timeline. Comparisons of the basic physical and chemical characteristics of different materials, finishes, and suppliers will be made relative to the requirements for implantable devices, high frequency RF devices, and portable electronics.
Why This 42-Step Medical PCB Guide Aligns Engineering Parameters and Procurement SLAs
An anonymized industry case involving a cardiac monitor showed how PCB trace defects can contribute to intermittent loss of signal during patient monitoring. The manufacturer’s exposure in such an event can be substantial and include device retrieval, lot isolation aligned with the finished-device manufacturer’s QMSR obligations, production line re-qualification, and regulatory reporting.
The failure was avoidable. In this type of event, the root cause can often be traced back to the lack of a Design for Manufacturing (DFM) review prior to the commencement of manufacture. The DFM is one of eight critical gates within the process flow, which separates high-yield medical product manufacturing from those susceptible to product failures in the field.
This guide consists of three things. Firstly, the complete step-by-step medical-grade PCB production process (42 total steps), re-organized around the eight DFM gates that capture the major causes of yield failure. Secondly, verification tables from the views of both the design and purchasing teams at each gate, so that both teams are utilizing the same information. Lastly, comparison tables covering equipment selection, substrate material selection, IPC Class 3 inspection, and surface finish selection, as well as a 2026 regulatory compliance framework covering FDA SPDF Cybersecurity, EU CSRD Carbon Disclosure, and DHR Traceability. Prior to visiting this area of research, there are two common assumptions about how the medical industry has defined its manufacturing requirements. Firstly, that step count is a sufficient measure of whether a manufacturer’s critical quality gates are present. Secondly, that Artificial Intelligence (AI) has sufficiently advanced to cover all design-for-manufacturing shortcomings medical boards will face. We will discuss both of these assumptions in more detail below.
Why Step Count Is the Wrong Metric for Medical PCB Quality
The yield of a medical-grade PCB manufacturing facility will be determined not by how many steps are listed on a supplier’s process flow, but by the presence of all the critical quality gates at each completed manufacturing lot — so that they can be staffed and verified by the manufacturer through multiple lots during the product life cycle.
The supplier process flow listing can contain anywhere from 20 to over 50 different steps; when purchasing teams utilize the total number of steps as a measure of sophistication, it does not accurately reflect how many quality gates have been established, or how many quality gates are verified.
Evaluating the actual defect data back to the initial causes of defects will reveal a pattern of clustering of defects for each of the eight defined process checkpoints. Therefore, a facility with 50 process steps and no plasma desmear capability will produce boards that have a shorter life than the same boards produced at a facility with 30 total production steps that has included the plasma desmear process within the scope of its total process and has evaluated that process for all lots produced.
The checkpoints with the largest impact on yield are the most significant within the medical PCB industry. These checkpoints include areas in which AI-driven DFM reviews can still miss important risks on both the inner and outer layers of a finished board, while a combined AI-and-engineer review can reduce scrap more effectively. As shown by defect data collected during manufacturing operations aligned with IPC-A-600H acceptance criteria, parts manufactured with only a standard AI-driven DFM review can show materially higher scrap than parts manufactured with both AI and engineer reviews running simultaneously.
In addition to the defect rates associated with conducting AI-driven DFM reviews, plating performance also varies by process architecture. In practice, products manufactured on conventional horizontal plating lines can show materially higher void risk than comparable products manufactured on well-controlled VCP lines. Several of the eight process checkpoints account for a disproportionate share of the yield difference between facilities that execute these checkpoints consistently and those that do not. The mSAP patterning process, the plasma desmear process, the alignment of the three-dimensional X-ray process, the verification of TDR (Time-Domain Reflectometry) impedance testing, the via-in-pad fill process with resin, and ionic cleanliness testing represent distinct mechanisms within a manufacturer’s process. Together, the eight processes provide a more robust framework for defining successful outcomes than the raw number of process steps listed in a supplier’s flow.
Given these realities, the clear implication to procurement teams is: “Do Not compare step counts in suppliers’ brochures!” Rather, require suppliers to provide documented evidence of the execution of critical quality gates — indices of process-capability, models of equipment used, and per-lot test results associated with each of the eight gates.
One of the greatest mistakes, however, that procurement teams are making is that they are not only using step count to compare suppliers, but additionally, are considering that DFM tools that are powered by AI technologies are advanced sufficiently to identify the full range of risks associated with the design of a PCB. The next section of this article will provide more details concerning where the DFM workflow stops — and what can happen as a result of not addressing the gap that will exist after the DFM workflow has been executed.
Where Standard DFM Workflows End and Senior Engineers Must Step In
An AI-driven review of Design for Manufacturing (DFM) is capable of detecting a meaningful share of the most common design rule infractions in HDI medical boards before a panel goes into production. However, current workflows for DFM do not incorporate the analysis techniques associated with full-wave electromagnetic simulation that can identify some of the causes of recalls for medical devices. Although this type of analysis can be performed using signal integrity-specific applications (e.g., Simberian, Ansys HFSS, Keysight ADS), these tools are not included in the typical DFM review workflow.

Dedicated signal integrity professionals are needed to perform these analyses using a different job environment, which creates an additional review step that most DFM workflows do not include as part of their default process.
The current generation of DFM engines (Valor NPI, Altium AI, Zuken CR-8000) performs exceptionally well in detecting simple geometric errors—like trace-to-copper spacing below the minimum design rule; insufficient annular rings flagged against IPC-6012E §3.3; solder mask aperture mismatches; and minimum via-to-pad separations. All of these are binary checks (i.e., each geometric placement either passes or fails), and AI performs them more rapidly than any human reviewer would be able to.
The four categories of design risk that are routinely overlooked as part of a standard DFM review process illustrate the gap between geometric errors and design risks. Signal integrity coupling at higher frequencies requires TDR simulation alongside stackup-specific Dk/Df modeling. The success of this analysis depends on the designer’s understanding of how the specific laminate being used will behave at various frequencies. In order to analyze the thermomechanical stress caused by mismatched CTE values during the lamination of higher-layer HDI PCB stackups, it is necessary to perform a sequential lamination cycle analysis of the Z-axis CTE mismatch. To identify the potential hazards from barrel cracking of via geometries, it is necessary to look at the interaction of these components with the resin system and the thermal profile; therefore, there is no existing DFM rule library available that covers all of these parameters. When designing a rigid-flex PCB assembly, it is important to ensure an impedance transition match between two different dielectrics at their respective material boundaries. To validate controlled-depth back-drilling techniques, the drill-depth tolerance must be cross-referenced against the actual copper-layer locations after the final lamination process.
Published DFM process data consistently shows that first-pass success rates are substantially stronger for simpler low-layer boards than for higher-layer HDI configurations with mixed dielectric materials. That gap correlates directly to the four categories mentioned above.
For medical-grade PCB manufacturing processes, the gap created by the difference in success rates between these two groups of boards represents a risk to patients. For example, a design having a signal-integrity defect that passes standard DFM screens can generate electromagnetic interference on a higher-layer imaging processor PCB. This electromagnetic interference can degrade the ultrasound images produced by the imaging processor; this type of failure often appears only during clinical validation after the product has been built. By that time, the cost to correct the defect is no longer a simple re-spin; it may require a corrective action or removal under 21 CFR Part 806, triggered by the finished-device manufacturer’s QMSR obligations under Part 820, with recall-related exposure that can become substantial depending on field volume.
An AI-driven first model and then an engineer’s second review methodology is employed. The AI automates the DFM screening of the entire Gerber data set against the geometric rule library and provides completed DFM files within 24 hours. Each PCB design exceeding 10 GHz operating frequency, 16 layers in number, any stacked microvia or any-layer HDI architecture regardless of layer count, a mixed dielectric stack-up, or classified as an implantable will undergo a second review by a named senior engineer considering the four categories of high-risk items. Both DFM screens yield signed documentation to be included as part of the Device History Record.
The following three criteria constitute the terms used by procurement teams writing the RFQs for this DFM model; suppliers must disclose the actual name of the AI DFM tool used and its documented catch rate for each project undertaken; a named senior engineer must approve every medical-grade HDI PCB and link the signature to a unique panel serial number; for 16-layer or larger HDI boards, the suppliers must define a dual review process for these products in their Quality Assurance Plan.
Now that both assumptions have been appropriately identified: 1) a step count is not an indicative measure of quality, and 2) there may be defined limits to how far one can go with standard DFM workflows, a solid foundation has been laid upon which to move forward. The next eight sections will sequentially describe the remaining critical process gates that comprise DFM review.
The Process Gates That Account for Most Medical PCB Yield Risk
The early steps of the 42-step production process for medical-grade PCBs provide an essential basis for all subsequent layers. While each step has its own parameter window, the DFM review gate is still the earliest indicator of whether the downstream process can support reliable Class 3 production.
How AI DFM Catches Common Defects (and Where It Fails)
According to medical PCB production experience, a common cause of production delays stems from recurring DFM (Design for Manufacturing) issues that can be identified by DFM software prior to imaging a panel. The majority of these issues are classified as “known issues”, meaning that these types of failures occur often but go unnoticed by the designer until it is too late to correct them.
The most common DFM issue is insufficient annular ring size for vias on the inner layers of printed circuit boards (PCBs). IPC-6012E Section 3.3 requires for Class 3 printed circuit boards (PCBs) a minimum annular ring size of 50 µm external and 25 µm internal. As soon as that annular ring is gone, the plated barrel does not have its mechanical anchor and will eventually become an open circuit after thermal cycling takes place.
The next most common DFM issue would be trace-to-copper spacing violations. These types of violations usually are related to solder mask aperture size and the size of the corresponding pads. The next most common DFM issue is plane-to-plane coupling that is not addressed by the design rules, resulting in improperly terminated impedances. These types of coupled planes cause issues that cannot be solved without making a re-spin of the printed circuit board (PCB).
The fourth most common DFM issue is the mixed dielectrics within stacked dielectrics. Most DFM libraries view all layers as homogeneous; therefore, they fail to flag them as mixed stack-ups.
The first three DFM issues are commonly addressed by AI detection methods. The remaining issues are outside the envelope of AI detection and require the use of electromagnetic simulation and material-specific frequency-response data, both of which are typically outside standard DFM workflows.
The process for capturing both DFM types is sequential. The AI engine scans through the entire Gerber file set and evaluates it for potential DFM issues. Once the AI has completed the scan, a senior engineer reviews the reported issues for the higher-risk categories of concern. Both the AI report and the engineer’s signature are then placed into the production record.
DFM Review Verification
| Verification Item | Engineering Specification | Documentation Requirement |
|---|---|---|
| DFM report turnaround | Target turnaround defined in QAP | Escalation trigger at 48h with documented root cause |
| AI catch rate | Documented standard DRC catch rate | Catch-rate report delivered per project |
| Senior engineer sign-off | Required for ≥16L HDI | Named engineer signature linked to panel serial |
| Mixed stackup review | Manual for Rogers/PI/FR4 hybrid | Documented in Quality Assurance Plan |
| SI coupling review | Required for higher-frequency signals | SI simulation report from dedicated tool |
| Thermo-mechanical review | Required for medical implantable | 1000+ cycle reliability prediction documented |
Discovering design flaws is just one of the first hurdles in this step of the process. The next is whether or not the supplier will be able to assemble all of the finer elements needed in the design; this is where modified semi-additive patterning comes into play.
How mSAP Delivers Higher Density Than Traditional Subtractive Etching
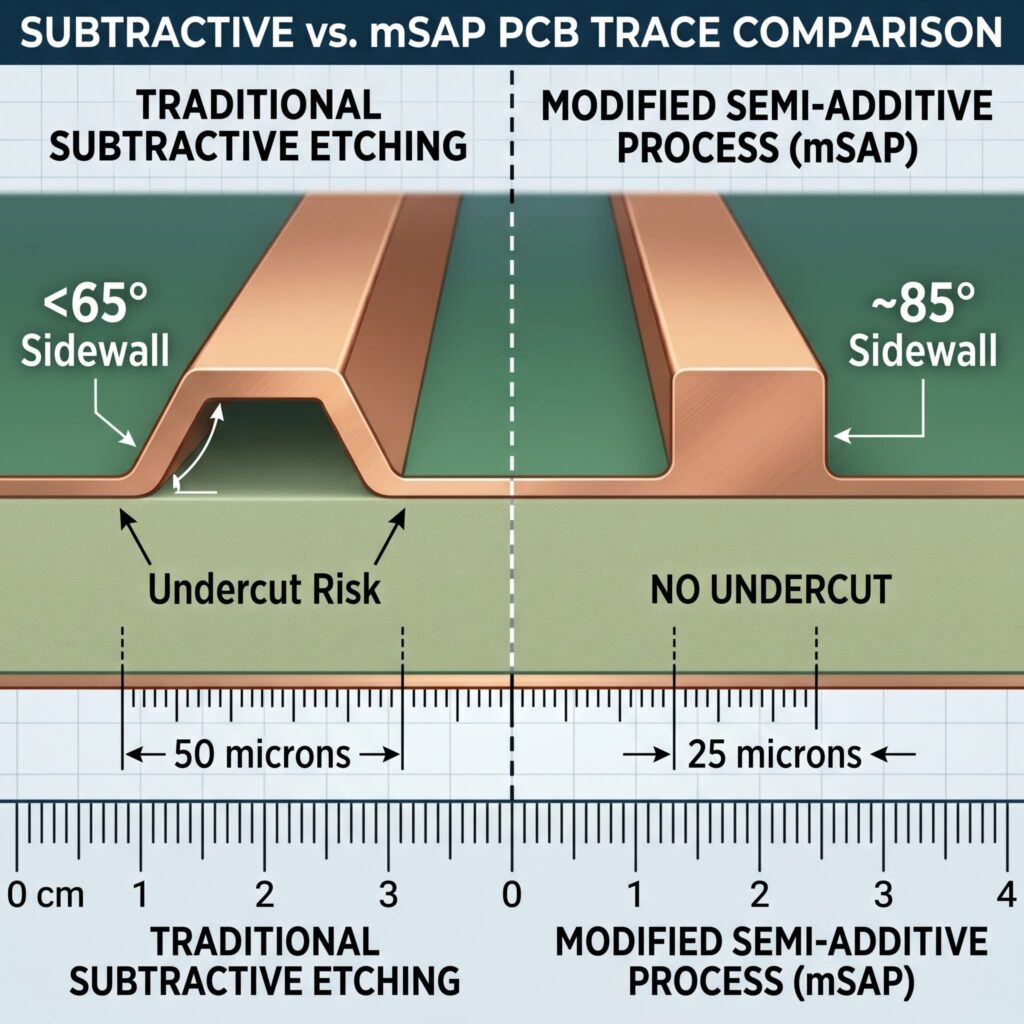
The 25/25 microns line/space resolution of manufacturing-proven medical HDI volumes using the mSAP process + the +15 microns/15 microns capabilities demonstrated with advanced platform.
The traces in the mSAP process are formed upwards using selective plating instead of the traditional method of etching downwards from bulk copper, thereby avoiding the undercut problem associated with subtractive processes, which can only produce approximately 50/50 microns traces in volume production.
The mSAP process has three stages, with each stage exhibiting its failure signature. The stages of the mSAP process are as follows:
The first stage is the deposition of the seed layer. A conductive film of 0.5-1.0 microns is deposited uniformly across the entire surface of the panel using an electroless copper bath. The seed thickness must not vary more than ±10% across the panel in order for the subsequent process of electrolytic plating to produce uniform trace widths that meet the ±5% tolerance required for controlled impedance.
Image plating is the second stage of the mSAP process. The photopatterned image created by the Laser Direct Imaging (LDI) process, which has a registration accuracy of ±5 microns as reported by the manufacturer on machines such as the Orbotech Ultra Dimension™. The photoresist image only exposes the locations of the traces; the electrolytic copper fills the exposed areas to a thickness of 15-25 microns above the seed layer. Because electrolytic copper is only deposited where it is needed, the side wall angle of the traces approaches 85° under optimized flash-etch conditions, compared to approximately 65° for traces produced by subtractive methods.
The flash etching stage is the removal of the seed layer between the traces. This stage has a time limit of less than three seconds, and if the removal process is continued for over that time, it erodes the underlying trace, which would then require more undercutting to create the trace, and that is exactly the design goal of the additive process. For medical-grade production, the threshold requirement for statistical process control (Cpk) during a flash etching operation is ≥1.33.
Three failure modes can be associated with these stages. If the seed layer is not deposited uniformly, localized line width variation may occur. If too much time is allowed to elapse during flash etching, a large area of undercut may occur. Instability in the electroless copper bath can generate voids in the fine-line traces, resulting in a reduction of the trace’s mechanical strength during temperature cycling.
mSAP Process Verification
| Verification Item | Engineering Specification | Documentation Requirement |
|---|---|---|
| Min L/S capability | Fine-line medical HDI capability | Certified by test coupon per lot |
| Line width tolerance | Controlled line-width tolerance | SPC chart delivered monthly |
| Seed layer uniformity | Tight seed-layer uniformity across panel | Cross-section report per lot |
| Flash etch SPC | Cpk ≥1.33 | SPC data shared quarterly |
| mSAP equipment vintage | ≤5 years | Equipment list provided during audit |
The following table benchmarks the core equipment a 2026 medical-grade facility should have in place across all critical process stages.
2026 Medical-Grade PCB Core Equipment Selection
| Process Stage | Equipment Model (2026) | Technology Type | Rated Tolerance (Mfr. Spec) | Key Benefit |
|---|---|---|---|---|
| Circuit Imaging | Orbotech Ultra Dimension™ | LDI (Laser Direct Imaging) | ±5 μm registration | Micron-level trace resolution for medical HDI |
| Hole Processing | Mitsubishi GTW4 Series | UV/CO2 Hybrid Laser Drill | 50 μm min. hole dia. | Consistent blind via formation in HDI stackups |
| Hole Metallization | Atotech Uniplate® InPulse2 | Horizontal Pulse Plating | >20% Cu elongation | High-ductility copper deposition for demanding PTH plating |
| Electrical Testing | Hioki FA1817 | Flying Probe Tester | 0.1 pF sensitivity | 100% netlist verification per IPC-9252 |
| Final Inspection | Mania AVI System | AVI (Automated Visual Inspection) | Pixel-level compare | Eliminates Class 3 cosmetic & functional defects |
| Layer Alignment | Automated 3D X-Ray Registration System | 3D X-Ray Alignment System | ±50 μm layer-to-layer | Zero misregistration in ≥16L HDI builds |
| Plasma Cleaning | March Plasma PX-250 | RF Plasma Desmear | <1% post-drill residue | Low-residue cleaning support for demanding medical boards |
| Etch Control | Schmid Group InlineTech | Differential Etching Line | ±2% etch uniformity | Fine L/S control down to 50/50 μm |
| Plating Control | VCP Line utilizing MacDermid Alpha or equivalent chemistry | Continuous Vertical Plating | Hole wall Cu ±3 μm | High AR uniformity for backdrilled vias |
Note: The tolerance values specified by the manufacturers are reflective of an ideal testing environment. However, due to manufacturing-controlled variables (like the quality of the production process), the production performance may vary based on these controlled variables, and other aspects such as maintenance schedules of the manufacturing equipment, and type of raw materials could also contribute to the variation in production performance.
The next production process gate evaluates if the production process produces a uniform thickness of copper inside the most vulnerable parts of the PCB, specifically the plated through holes designed to accommodate larger aspect ratio through-holes than what was available with traditional PCB manufacturing methods.
Why VCP Improves High-Aspect-Ratio Uniformity
Manufacturer literature for Vertical Continuous Plating (VCP) indicates that VCP can provide tighter copper-thickness uniformity through high-aspect-ratio plated through-holes (PTHs) than traditional horizontal plating designs, largely because of how current density is distributed throughout the hole.
When using horizontal plating lines, there are restrictions on the exchange of electrolyte at the center of the high aspect ratio hole due to the construction of the hole. For example, 0.3 mm holes drilled through 3.6 mm thick boards would receive copper ions into the hole from the edge of the board relatively easily, but due to the thickness of the substrate, the copper ions would be depleted before reaching the center of the hole. As a result of this phenomenon, the copper deposited at the wall of the hole creates a “dog bone” effect; therefore, while there is sufficient copper near the hole entrance and knee area, there is an inadequate amount of copper at the center of the barrel where the wall is thinnest, and it will be below the IPC-6012E §3.6 minimum requirement of 20 μm for Class 3.
Horizontal pulse plating systems have reduced some of these issues in moderate aspect-ratio applications; the impulse reverse waveform used in horizontal pulse plating periodically removes excess copper from each hole entrance and helps even out deposition along the length of the wall. For less demanding aspect-ratio designs, horizontal pulse plating can still offer an effective, lower-cost alternative method of plating holes.
For more demanding high-aspect-ratio circuits, the architectural benefits of VCP typically prevail. With the panel hanging vertically, gravity assists in the escape of the electrolyte from the hole. VCP also utilizes dedicated eductor nozzles that provide a consistent flow of solution through each hole at a controlled velocity. In addition, the segmented anode rails of the VCP process reduce current crowding at the entrance of the hole and move more of the current energy toward the midpoint of the barrel where copper depletion is greatest. By contrast, the Atotech Uniplate® InPulse2 system is marketed for high-ductility copper deposition, which is relevant to whether the plated barrel can endure the thermal expansion associated with lead-free reflow.
When neither VCP nor advanced horizontal pulse plating is used for demanding high-aspect-ratio circuits, plating voids can become a meaningful share of total defect volume, according to supplier process literature. Voids that form during the plating process concentrate at the midpoint of the barrel; therefore, such voids cannot be seen using surface automated optical inspection (AOI) and can only be identified using destructive microsections or X-ray computed tomography (CT).
High-AR Plating Verification
| Verification Item | Engineering Specification | Documentation Requirement |
|---|---|---|
| PTH AR capability | Validated high-AR capability for medical HDI | Test coupon delivered per lot |
| Wall Cu uniformity | Tight wall-copper uniformity (mfr. spec) | Microsection report monthly |
| Void rate | Low void rate with SPC | SPC chart with per-lot data |
| Bath management | Automated additive dosing | Equipment specification in audit file |
| Plating method for AR >10:1 | VCP or validated pulse plating | Process qualification report |
The copper deposit should distribute uniformly throughout the barrel, but the overall integrity of the barrel is still dependent on the condition of the area between the copper and resin along the edge of the hole where they meet (the hole-wall interface). If any residual material (drill smear) remains in the barrel prior to plating, it will result in an imperfectly uniform copper deposit where the drill-smear residue is present.
Why Plasma Desmear Removes Residue More Reliably Than Chemical Methods
Plasma desmear can reduce residual resin left over from drilling to a much lower level than is typically achieved with standard chemical desmear, because it attacks the resin smear through more than one mechanism rather than relying on a single chemical pathway.
Chemical desmear requires the use of a potassium-permanganate-based solution for dissolving the resin smear via a strictly chemical action. While effective in removing resin smear from accessible surfaces, there are limitations to how well a permanganate solution can enter and dissolve resin smear at the bottom of a blind via in higher-aspect-ratio structures.
On the other hand, plasma desmear works within a vacuum environment using radio-frequency (RF)-excited gas. It has been shown that a combination of physical sputtering and chemical reaction mechanisms work together during plasma desmear to remove resin smear. The physical sputtering of the resin occurs when ion bombardment from the RF-excited gas transfers enough kinetic energy to the resin molecules to eject them from the via site. The second mechanism is the creation of reactive oxygen species during the plasma desmear process, converting the resin polymer to volatile carbon dioxide (CO₂) and water (H₂O). Both methods operate in a gaseous phase, which means cleaning efficiency is less constrained by via geometry than in purely wet-chemical approaches.
The composition of the residue in an implantable application is just as important as the quantity of it. The chemical structure of potassium permanganate introduces both potassium and manganese ions into the via structure as contaminants. Once installed and under implant operating conditions (sustained DC bias, 37°C body temperature, and physiological moisture), both of these contaminants become the feedstock for allowing the development of electrochemical migration (ECM) failure modes. These failure modes typically develop over time with long-term exposure to conditions created in an implant.
Based on the threshold for Ionic Cleanliness (1.56 μg NaCl eq./cm²) outlined in IPC-J-STD-001 Class 3, even though this threshold continues to be referenced, it is a historical benchmark for the classification and reference of the connectivity of a product. In the present day, the electronics industry has adopted an Objective Evidence Based (OEB) approach to cleanliness determination with respect to the ionic profile of a product rather than a single concentration reference threshold. Therefore, the acceptance criteria for implantable boards should not be considered a fixed threshold. Instead, the acceptance criteria should be established either through OEB analysis or ion chromatographic analysis that takes into account the operating conditions, bias conditions, and the expected service life of that specific device.
Desmear Process Verification
| Verification Item | Engineering Specification | Documentation Requirement |
|---|---|---|
| Desmear method | 100% plasma for medical boards | Written process guarantee |
| Residue level | <0.5 μm residue thickness post-desmear (per microsection, IPC-TM-650 §2.1.1) | Per-lot microsection report |
| Plasma equipment | March PX-250 or equivalent | Equipment list in audit file |
| Cleanliness standard | OEB analysis for implantable; legacy 1.56 μg for Class 3 | Documented cleanliness protocol in QAP |
The DFM review, mSAP patterning, high-AR plating, and plasma desmear gates account for a large share of the yield risk during the production of medical PCBs (Printed Circuit Boards). The final four gates transition from fabricating layers to verifying the PCB at the board level, beginning with layer-to-layer alignment.
Verification Gates That Address the Remaining Yield Risk
Having completed the first four process gates, we have now verified that our production line has provided layers with correct geometry and fine-line resolution, as well as a uniform barrel copper surface with no contamination. By completing the following fabrication and inspection steps, we PASS or FAIL the layers based on quality checks such as inner-layer AOI (Automated Optical Inspection), continuity testing, oxide treatment, lay-up registration, vacuum lamination, edge trimming, drilling, microvia formation, and post-drill plasma cleaning. The final gate checks whether the entire multilayer stackup remains within specification.
How 3D X-Ray Alignment Achieves IPC-6012 Class 3 Layer Registration
The IPC-6012E §3.4 standard specifies a true-position registration tolerance of ±100 μm for Class 3 medical PCBs (printed circuit boards) relative to each other. PCB manufacturers often tighten this tolerance for higher-layer HDI stackups because the standard tolerance does not account for the accumulation of drift caused by multiple lamination cycles.
The more traditional optical registration methods used by PCB fabricators locate layer-to-layer alignment using the outermost layer fiducial markings. A simpler multilayer PCB manufactured during a single lamination cycle may still show acceptable correlation between those fiducials and the internal layer locations. Sequential lamination changes this relationship. A higher-layer HDI PCB can require multiple lamination cycles, each performed under elevated temperature and pressure, inducing lateral drift due to resin flow during cure.

As an example of this cumulative mechanical displacement due to multiple lamination cycles, the average offset allocated to the copper circuitry during cure can accumulate enough over several cycles to consume much of the available Class 3 registration budget.
Another issue experienced during HDI PCB manufacturing is varying degrees of CTE (coefficient of thermal expansion) mismatch between the copper-dominated signal layers and resin-dominated prepreg layers of the PCB. There are also locations of the copper circuitry where the CTE mismatch will create unequal amounts of dipole movement between prepreg and copper-laden materials, resulting in uneven positional shifts of the PCB’s copper features.
By employing a two-part 3D X-Ray system, such as the Automated 3D X-Ray Registration System (rated at ±50 μm), all of the layers will be penetrated and the copper circuitry located on all of the internal layers measured simultaneously and volumetrically. For each layer of a PCB, a comparison of its position against the known fiducial location will be made in real time and displayed as an error in registration. Thus, when measuring registration between layers, the registration errors can be quantified on a layer-by-layer basis as well as per panel.
Layer Alignment Verification
| Verification Item | Engineering Specification | Documentation Requirement |
|---|---|---|
| Layer registration (Class 3) | ≤100 μm per IPC-6012E §3.4 | 100% X-Ray verification per panel |
| 16L+ tightened spec | Tightened internal spec for higher-layer HDI | Per-lot microsection confirmation |
| X-Ray equipment type | 3D CT-based (not 2D) | Equipment model documented in audit |
| Sequential lamination count | Documented per design | QAP record per build |
Layer alignment confirms that all features sit where they belong. The next gate verifies that electrical signals traveling through those features maintain their designed impedance.
How to Set Up and Interpret TDR Impedance Testing for Medical High-Speed Boards
Time-Domain Reflectometry creates an impedance profile of each controlled-impedance trace on a finished board. For higher-speed medical devices, TDR is the primary production-floor method (complemented by VNA in frequency-domain analysis) to verify impedance discontinuities that would not be detected by electrical continuity testing.
Test Set-up
The accuracy of TDR depends on three parameters of the test equipment. First, the rise time of the instrument determines the spatial resolution. Per manufacturer specifications, very-fast-rise-time platforms provide the spatial resolution needed for small PCB features. Second, the signal coupling method affects the quality of the launch. SMA connectors used on dedicated test coupons provide the best possible launch; however, landing probe tips on finished boards is still an acceptable method for production monitoring because the higher launch impedance creates more uncertainty about the actual impedance of the traces being tested. Finally, temperature control of the test environment helps limit dielectric-constant drift that can compromise compliance with the IPC-2141A tolerance window.
Waveform Interpretation
A downward dip on an impedance vs. time plot at a via transition indicates that the via stub is adding capacitive loading. An upward step occurs when there is a narrowing of the coupled trace or reference plane gap. A gentle slope of the impedance vs. time over a long trace suggests a non-uniform dielectric thickness from a lamination process. Each of these signatures has a unique physical root cause associated with it, and can be traced back to a particular upstream process.
IPC-2141A specifies that controlled-impedance designs must maintain an impedance tolerance of ±5% for designs up to 10 GHz. For medical imaging systems such as ultrasound processors and MRI receiver coils and for surgical navigation sensors, a mismatch in this tolerance range will return reflected energy back into the signal path as noise and could have a clinical impact that will necessitate compliance with FDA design verification requirements pursuant to 21 CFR Part 820.
Impedance Verification
| Verification Item | Engineering Specification | Documentation Requirement |
|---|---|---|
| Impedance tolerance | Controlled-impedance compliance per design | Per-design coupon test report |
| TDR rise time | <9 ps (mfr. spec) | Equipment specification in audit |
| Coupon design | IPC-2141A compliant | Design review sign-off documented |
| Test report delivery | 100% tested designs | Report delivered per lot |
TDR offers assurance that the signal will propagate through the proper impedance. A medical HDI design with via-in-pad construction will have a signal pathway that goes through a filled via, and if the filled via is not maintained in good condition, the impedance profile will not remain intact after experiencing a maximum of 1,000 thermal cycles.
How to Verify Via-in-Pad Resin Fill Meets IPC-4761 Void-Free Requirements for Medical Implantables
IPC-4761 Type VII is the via-fill classification commonly specified for highly demanding medical applications. In dense multilayer boards with fine-pitch BGAs, via-in-pad construction often becomes unavoidable. The process used to fill, planarize, and cap a via occurs in multiple steps and creates a unique failure mode at each step of manufacture. Filling the via requires that the resin be thermally matched to the substrate and drawn into the via under vacuum. The cure profile also determines the level of cross-link density achieved in the cured resin. If the resin is not held sufficiently long at peak temperature, the cured resin will be mechanically weaker and allow moisture to penetrate. Dimple depth must also remain controlled so that the secondary copper cap does not become locally thin.
Under implant conditions, the coefficient-of-thermal-expansion (CTE) mismatch between the resin fill material and the copper barrel creates Z-axis stress at the boundaries of any voids, causing cracks to initiate and propagate through the void over repeated thermal cycling, which can eventually separate the barrel connection. Moisture entering through any micro-cracks allows ionic migration — the same mechanism that drives electrochemical migration and that makes the plasma desmear and ionic cleanliness gates important.
Why a single verification method cannot provide full confidence
X-Ray CT provides full 3D mapping of voids within a PCB, while metallographic microsectioning is a destructive test that shows void fraction, dimple depth, cap thickness, and interface quality in a single image. C-SAM is a non-destructive verification method but is less sensitive to smaller defects. Therefore, for PCBs intended for implantable devices, the practice standard for verification is to combine X-Ray CT with microsectioning so that volumetric imaging is supplemented by destructive confirmation.
Via Fill Verification — Implantable Grade
| Verification Item | Engineering Specification | Documentation Requirement |
|---|---|---|
| Via fill type | IPC-4761 Type VII | Written process guarantee |
| Void acceptance | Within IPC-4761 Type VII guideline | CT scan report per lot |
| Dimple depth | ≤75 μm post-planarization | Microsection report per lot |
| Resin material | Low-CTE thermal match | Material datasheet in audit file |
| Cure profile | Documented oven profile per resin spec | QAP record |
| Secondary plating cap | Cu ≥25 μm per IPC-6012E §3.6 | Thickness measurement per lot |
| Thermal cycling validation | 1,000+ cycles for implantable | Reliability test report |
Via fill integrity ensures reliable Z-axis interconnects. The final gate addresses the last contamination check before the board leaves the production line.
Why Ionic Cleanliness Testing Is Non-Negotiable for Medical Boards
The importance of this change is that medical device applications vary dramatically with respect to their ionic-contamination risk profiles. For instance, a portable diagnostic device is typically used in an ambient room-temperature environment, whereas an implanted neurostimulator operates at body temperature in physiological saline with a sustained DC bias applied on the device.
The legacy standard of 1.56 μg NaCl equiv./cm² was designed as a broad historical benchmark, whereas the OEB methodology allows the appropriate threshold to be defined for each product category based on its actual use conditions.
For this reason, there are three test methods available, each serving a different purpose. ROSE testing is a fast, broad-based technique that averages contamination across the entire board but provides no information regarding localised hot spots. Ion chromatography (IC) identifies, separates, and quantifies the individual ionic species to provide a means for root-cause analysis. Surface insulation resistance (SIR) testing, according to IPC-TM-650 §2.6.3.7, uses accelerated environmental conditions to observe the failure mechanism. For implantable devices, SIR validation helps demonstrate that accelerated aging does not produce measurable leakage paths.
Final Contamination Verification
| Verification Item | Engineering Specification | Documentation Requirement |
|---|---|---|
| Ionic contamination | Per OEB analysis or legacy 1.56 μg/cm² | Per-lot IC test report |
| Implantable threshold | OEB-determined product-specific limit | Documented OEB protocol in QAP |
| Test method | IC required; SIR for implantable qualification | Method documented in QAP |
| Test frequency | Every 24h of production | SPC chart with trending data |
With all eight process gates mapped, the quality framework is complete. But even with rigorous gate execution, failures still occur. The next section dissects the five most common yield killers, tracing each from symptom through root cause to fix.
Common Medical PCB Yield Killers — From Symptom to Root Cause to Fix
Eight gates in the manufacturing process significantly decrease the likelihood of defects occurring. If failures do occur, the speed at which the engineering team can troubleshoot depends on their ability to identify the symptom and trace it back to the specific process parameter that is out of spec. The following scenarios represent common yield killers.
Failure 1: Inner Layer Scrap From Incomplete DFM Review
In a new higher-layer HDI design, inner-layer AOI flagged a meaningful share of scrap due to shorting problems after etching and trace-to-trace shorts in a densely populated area under the BGA fan-out. Although the spacing met the geometric requirements of DFM, the AI did not evaluate the effect of electromagnetic coupling, which is outside the standard DFM workflow. The data indicate a high level of crosstalk at tight spacing on a thin dielectric at elevated frequency. Solution: implement a dual review process consisting of AI and engineer review for higher-speed designs. Preventive gate: a signed dual DFM report in the production record.
Failure 2: Layer Misregistration in 16+ Layer Builds
The flying probe test identified intermittent opens on a higher-layer board. The coupons exhibited registration close to the IPC-6012E §3.4 limit. The stack-up process created cumulative drift that could not be detected by optical alignment alone. Resin flow during multiple lamination cycles induced additional movement, eventually causing annular-ring breakout on the inner layers. Solution: a 3D X-ray registration process with tighter control than conventional optical alignment. Preventive gate: full X-ray verification for each panel.
Failure 3: Plating Voids in High-Aspect-Ratio Vias
Kelvin testing of a high-aspect-ratio via showed evidence of intermittent high resistance. Microsectioning showed mid-barrel copper below the required Class 3 minimum. The horizontal plating configuration did not allow sufficient electrolyte exchange, resulting in copper depletion at the mid-barrel location. Combined fix: validated pulse plating (or VCP) for demanding aspect-ratio designs with real-time current-density monitoring. Preventive gate: routine microsectioning with SPC by lot on void rate.
Failure 4: ENIG Black Pad on BGA Components
BGA parts do not wet during reflow. Microsectioning indicates hyper-corrosion of the nickel layer; phosphorus enrichment at the nickel-gold interface caused aggressive grain-boundary attack during the immersion gold application. The corroded grain boundaries cannot form an inter-metallic bond to solder. Scrap all affected boards. Combined fix: ENEPIG per IPC-4556A for implantable applications; the palladium barrier helps block the galvanic-displacement mechanism. Preventive gate: regular XRF coupons with SPC on phosphorus content and bath control.
Failure 5: Via Fill Void in Implantable Board
Reliability testing discovered intermittent opens after extended thermal cycling on a cardiac-device board. CT scanning showed void area beyond the IPC-4761 Type VII guideline. A vacuum drop during the filling process allowed air to remain trapped in multiple vias. The CTE mismatch between the resin and copper then concentrated stress at the void boundary over repeated cycling. The lot is on hold pending regulatory review. Combined fix: continuous vacuum monitoring, automatic stop logic, and CT scanning by lot for implantable via-in-pad boards. Preventive gate: CT plus microsectioning dual verification, along with preventive maintenance of the vacuum system.
All these situations are a function of a specific process gate or a specific parameter drift. The next question is what to require in selecting between the competing materials, finishes, and suppliers for a specific application in medicine.
Medical PCB Supplier Selection Framework — Use Case Scenarios
Scenario 1: Implantable Class 3 — Pacemakers, Neurostimulators, Cochlear Implants
Continuous body temperature, saline exposure, sustained DC bias, long service life, and repeated thermal cycling make implantable boards the most demanding use case. The process gates that are most critical are the contamination and fill-integrity gates, specifically plasma desmear, via-in-pad resin fill, and ionic cleanliness, because the operating environment creates more opportunity for electrochemical migration and via-barrel fatigue than any other product type. The substrate type used will depend on the RF telemetry section, either Shengyi S1000-2M (halogen-free, high-Tg) or Rogers RO4350B (a common RF laminate per the manufacturer’s datasheet). The surface finish will be ENEPIG per IPC-4556A.
Scenario 2: High-Frequency RF Medical — Ultrasound, MRI Coils, Telemetry Monitors
This type of application is expected to involve elevated signal frequencies. These products are typically used in a controlled environment and remain in service for multiple years with several potential reflow cycles. The gates that are of primary concern are those associated with signal integrity: mSAP patterning, high-aspect-ratio plated structures, and time-domain reflectometry (TDR) verification. The types of substrate material used in this type of product would either be Isola Astra MT77 for lower loss or Taconic TLX-8 for lower dielectric constant, according to the manufacturers’ datasheets. Standard FR-4 can become less suitable as frequency increases because of its higher loss tangent. Surface finish will typically be ENIG by default and ENEPIG when assembly demands are higher. The core manufacturing requirements for this type of application are ISO 13485 and IPC-6012 Class 3, while FCC Part 15 applies at the finished-device level.
Scenario 3: Portable Diagnostic — Point-of-Care Ultrasound, Holter, Wearable Cardiac
These products typically experience intermittent use, transportation vibration, and a shorter service life than implantable or fixed-system boards, and they are often more cost-sensitive. In this category, DFM reviews and 3D X-ray alignment are weighted more heavily than some of the other gates. Most boards produced for this application are typically moderate-layer-count constructions. The substrate materials will either be Panasonic Megtron 7 or Isola I-Tera MT40 according to the manufacturers’ datasheets. The surface finish will be ENIG per IPC-4552A. Minimum certification for this type of product is ISO 13485, but IPC-6012E Class 3 is still highly recommended.
Use Case Mapping
| Application Scenario | Priority Process Gates | Preferred Substrate | Surface Finish | Required Certifications |
|---|---|---|---|---|
| Implantable Class 3 | Plasma / Via Fill / Ionic | Shengyi S1000-2M / Rogers RO4350B | ENEPIG | ISO 13485 + FDA + ISO 10993 |
| High-Freq RF >6 GHz | mSAP / High-AR Plating / TDR | Isola Astra MT77 / Taconic TLX-8 | ENIG → ENEPIG | ISO 13485 |
| Portable Diagnostic | DFM Review / X-Ray Alignment | Panasonic Megtron 7 / Isola I-Tera | ENIG | ISO 13485 |
High-Performance Medical-Grade Substrate Material Comparison
| Brand / Series | Material Grade | Tg (°C) | Dk @10 GHz | Df @10 GHz | Medical Application | CTE Z (ppm/°C) |
|---|---|---|---|---|---|---|
| Panasonic Megtron 7 | Ultra Low Loss | 215 | 3.6 | 0.002 | Imaging processor boards | 12 |
| Isola I-Tera MT40 | High-Speed Digital | 200 | 3.45 | 0.0031 | Portable ultrasound | 15 |
| Rogers RO4350B | RF/Microwave PTFE-free | >280 | 3.48 ±0.05 | 0.0037 | Wireless RF front-end | 11 |
| Shengyi S1000-2M | High-Tg Halogen-Free | 175 | 4.6 | 0.013 | Implantable monitoring | 16 |
| Taconic TLX-8 | Low Loss RF Thermoset | 200 | 2.55 ±0.04 | 0.0019 | MRI RF receive coils | 14 |
| DuPont Pyralux AP | Flexible Polyimide | 260 | 3.1 | 0.004 | Wearable cardiac flex | 20 |
| Isola Astra MT77 | Very Low Loss Hybrid | 170 | 3.0 | 0.0017 | Surgical navigation | 13 |
Note: Dk/Df values referenced are taken from manufacturers’ data sheets based on specific testing conditions. Please confirm against updated current-year datasheet revisions before specifying any material in production.
Red Flags — When to Walk Away
The absence of ISO 13485 certification may disqualify a supplier. A supplier that does not have a documented plasma desmear process cannot guarantee the amount of residue remaining in the implantable. If a supplier continues to use basic horizontal plating with an aspect ratio >10:1 and does not have pulse or VCP capabilities, there exists a capital gap. A supplier without CT-scanning capability is disqualified from implantable via-in-pad programs. A supplier’s refusal to provide microsection data for each lot indicates undetected variability due to production problems.
RFQ Integration
Each medical PCB RFQ must have three technical requirements: a documented stack-up published with the materials indicated by the substrate comparison table; controlled impedance test coupons per IPC-2141A; process-gate-specific verification coupons (one for each process gate). In order for each of these documents to be legally enforceable, three terms must be included in the contract: DFM report to be delivered within 24 hours; per-lot quality records must be sent with each shipment; and nonconformance reports must be provided within 48 hours along with a root cause analysis.
Decoding 2026 Medical PCB Standards — IPC Class 3, FDA SPDF, and IEC 81001-5-1
PCB manufacturers often mention IPC Class 3, ISO 13485, and FDA registration (where applicable as a component supplier to a registered OEM, since 21 CFR Part 820 generally exempts component manufacturers per §820.1(a)(1)) in their brochures. However, very few provide practical detail on what these standards require at the process-parameter level for successful product execution. For medical applications, procurement documents may also specify IPC-6012EM in addition to the base IPC-6012 requirements.
IPC Class 2 vs Class 3 — What the Classification Actually Requires
Class 3 IPC defines three classes of products based on their end-use reliability: Class 2 – Dedicated Service Electronics; Class 3 – High-Reliability Electronics. Therefore, the distinction between Class 2 and Class 3 is comprised of a series of binary thresholds; the annular ring for Class 3 per IPC-6012E §3.3 (superseded by IPC-6012F in October 2023; §3.3 requirements remain consistent) is: External – 50 μm minimum; Internal – 25 μm minimum (for Class 2, there is a 20% reduction); the layer registration for Class 3 by IPC-6012E §3.4 is ±100 μm; Class 2 allows ±125 μm; the rework section of the IPC-A-610, Section 8, for Class 3 is 0 (zero) repairs in critical areas; Class 2 allows 2 repairs. All Class II and Class III medical devices, as well as life-sustaining and implantable devices (sub-category Class 3/A), are required to be manufactured in accordance with IPC Class 3 standards.
IPC Class 3 Medical-Grade Inspection Protocol
| Inspection Feature | Class 3 Requirement | Standard Reference | Monitoring Frequency | Detection Method | Failure Consequence |
|---|---|---|---|---|---|
| PTH Cu Thickness | Avg ≥25 μm; min ≥20 μm | IPC-6012E §3.6 | Per lot microsection | Cross-section + SEM | Barrel crack under thermal cycling |
| Conductor Defects | No solder repair; zero rejectables | IPC-A-610 Class 3 §8 | 100% panel inspection | AOI + manual microscope | Signal loss, EMI radiation |
| Surface Finish (ENIG) | Ni 3.0–6.0 μm / Au ≥0.05 μm | IPC-4552A Rev. A | Weekly XRF coupon | XRF Spectrometer | BGA non-wetting, black pad |
| Ionic Contamination | Per OEB or legacy 1.56 μg/cm² | IPC-J-STD-001 Class 3 | Every 24h production | IC Tester | ECM, dendritic short |
| Layer Registration | ≤100 μm true position | IPC-6012E §3.4 | 100% X-Ray | Automated 3D X-Ray Registration System | Broken annular ring |
| Impedance Tolerance | ±5% @≤10 GHz | IPC-2141A | Per impedance design | TDR | Signal distortion, EMI |
| Solder Mask Adhesion | Class 3 tape peel | IPC-SM-840E §3.7 | Per panel/lot | Cross-hatch peel | Delamination under 260°C reflow |
| Min. Annular Ring | ≥50 μm ext; ≥25 μm int | IPC-6012E §3.3 | 100% AOI | AOI + microsection | Breakout, unreliable barrel |
Supplier Audit Checklist
| Audit Item | Requirement | Evidence Required |
|---|---|---|
| IPC-6012E Class 3 | Current certification | Certificate + latest audit report |
| IPC-A-610 Class 3 | Certified inspectors | CIT/CIS certification records |
| ISO 13485 | Active status | Latest surveillance audit report |
| FDA registration (if applicable) | Valid establishment | FDA registration ID or OEM traceability record |
| HBOM capability | Documented process | Sample HBOM output |
| IEC 81001-5-1 | Staff awareness | Training log with dates |
| DHR linkage | Defined process flow | QAP with panel serial mapping |
FDA SPDF and IEC 81001-5-1 — Cybersecurity at the PCB Level
In 2023, the FDA released the final version of its Secure Product Development Framework (SPDF) guidance, which includes a Refusal-to-Accept policy for premarket submissions that are not accompanied by cybersecurity documentation. IEC 81001-5-1:2021 provides a consistent international framework for SPDF, and as such, includes many aspects of IEC 81001-5-1:2021 within its content.
The requirements established in this framework are based upon four main physical characteristics of devices that are required to be in compliance; the central federal requirement is a Software Bill of Materials (SBOM) per FDA Section 524B(b)(3) covering commercial, open-source, and off-the-shelf software components in machine-readable format (SPDX or CycloneDX). A complementary Hardware Bill of Materials (HBOM) is increasingly requested by OEMs to support component-level traceability, though not federally mandated at the PCB level. Secure boot architecture, side-channel attack resistance, and firmware update paths are addressed at the finished-device design level rather than by the PCB supplier.
Supply chain considerations include the following: all suppliers must demonstrate the capability to generate HBOMs; demonstrate knowledge of how to create documented models of threats; and, in the case of European markets, be expected to meet the dual certification baseline of ISO 27001 and ISO 13485 by 2026.
21 CFR Part 820 (QMSR) — Device Record Traceability Linkage
The Device History Record (DHR) — now harmonized as the “Device Record” under the QMSR effective February 2, 2026, which incorporates ISO 13485:2016 §4.2.5 by reference and supersedes the pre-2026 standalone §820.184 structure — represents the link between device PCB production and the manufacturer’s regulatory records. The DHR should include raw-material lot numbers, a record of the process steps associated with panel serial numbers, the results of outgoing inspections, and the records of RFID-enabled packaging. In practice, the link between the DHR and the PCB is established through panel-level serialization and carried through the manufacturing record.
Medical PCB Procurement Lifecycle — From RFQ to DHR Traceability
This section consolidates the individual verification tables into a single procurement lifecycle framework mapping supplier requirements, documentation deliverables, and acceptance criteria across all ten production stages.
Stage-by-Stage Supplier Requirements
| Production Stage | Engineering Specification | Documentation Requirement | Escalation Trigger |
|---|---|---|---|
| Pre-production DFM | AI + senior engineer dual review | Signed DFM report | No report at 48h → escalation with root cause |
| Inner layer fabrication | AOI 100% + X-Ray | AOI log + X-Ray report | Any Class 3 rejectable → NCR within 48h |
| Lamination | 3D X-Ray alignment | Alignment verification report | Any layer >50 μm drift → hold for review |
| Drilling | UV/CO2 laser for microvias | Test coupon + microsection | AR capability not demonstrated → qualification hold |
| Plating | VCP or validated pulse for AR >10:1 | SPC chart per lot | Void rate >0.1% → process review triggered |
| Via fill (implantable) | IPC-4761 Type VII | CT scan report per lot | Void >10% → lot quarantine |
| Surface finish | ENIG or ENEPIG per scenario | XRF coupon log weekly | Ni/Au out of spec → batch hold |
| Ionic cleanliness | Per OEB or legacy threshold | IC chromatogram per lot | Threshold exceedance → lot quarantine |
| Final validation | TDR + cross-section | Test report per lot | Impedance >±5% → design review triggered |
| Packaging | RFID + vacuum lightproof | DHR record per shipment | Missing RFID data → shipment hold |
Device History Record Traceability at the PCB Level
In accordance with the FDA’s Quality System Regulation, a Device History Record (DHR) must be maintained for every production run of a medical device. PCB-level compliance is defined by four elements: material-lot traceability showing a direct relationship between incoming materials and their Certificates of Analysis (COA), process-step documentation establishing an association between test results and panel serial numbers, RFID-integrated packaging enabling traceability from shipment through receipt, and Manufacturing Execution Systems (MES) integration allowing OEM quality teams to query the records.
RFQs with four clauses will assist in achieving DHR compliance: each lot of material should be accompanied by a COA, panel-level serial numbers should be provided in both QR and RFID formats, data should be maintained for an appropriate retention period aligned with the quality system, and documentation of MES API capability should be provided as part of supplier qualification.
Supplier Evaluation Signals
A supplier that is not able to provide panel-level serial number traceability has no means of isolating a recall — which means that any field action will encompass the full history of production. A supplier with data retention less than three years is not able to fulfill the OEM’s obligation of maintaining their records for five years. Conversely, a supplier that has invested in a traceable system of RFID-integrated packaging, along with cloud-based MES and API access to its records, has demonstrated both a financial commitment to providing this level of service and, through the audit process, has demonstrated that it can provide the entire traceability from the panel back to the material lot in less than one hour during the qualification process.
Why Rigorous Process Gate Execution Delivers ROI — Including 2026 ESG Compliance Value
The investment rationale for implementing a gates-based method for managing the manufacturing process of medical devices financially is based on the differential between the cost incurred to prevent and the cost incurred due to the failure of a medical PCB in manufacturing.
The cost structure of medical PCB failure
The cost incurred to create a panel with the intent to detect issues during quality control is modest compared with the cost of a field failure in a medical application. If an implantable or life-supporting device fails, the cost of retrieval can affect multiple elements. For example, the finished-device manufacturer may need to notify FDA under 21 CFR Part 806 (corrections/removals), quarantine affected lots while audit data is reviewed, trace the full DHR, re-validate the production line, and potentially manage litigation exposure.
Yield differential.
Facilities operating with all eight quality gates verified and documented generally report stronger first-pass yields for medical HDI boards than facilities missing multiple gates in comparable builds. On meaningful production volume, even a modest yield improvement can translate into substantial direct material savings per lot before downstream costs of delay, quality escapes, and gate infrastructure are netted out.
Each gate investment has a discrete return profile. AI DFM with senior engineer review can reduce inner-layer scrap relative to AI-only review. VCP or advanced pulse plating can sharply reduce void risk associated with basic horizontal lines on high-AR via structures. Plasma desmear supports the cleanliness expectations required for implantable certification, unlocking a higher-margin product segment. CT scan capability also helps qualify a facility for implantable programs with stronger per-board margins.
2026 ESG compliance: market access, not cost center.
Several regulatory and market forces are making environmental sustainability a qualification factor in the procurement process. The EU Corporate Sustainability Reporting Directive will phase in starting in 2024, and as of 2026 many medical device OEMs that sell products into European markets will need carbon-footprint information from PCB suppliers for Scope 3 reporting. Without the ability to supply per-lot carbon-intensity metrics, a supplier may be unable to complete the OEM’s own CSRD disclosure and can become harder to qualify. The market has also created additional requirements at the material level to comply with RoHS 3.0 requirements (noting that medical devices retain specific exemptions under RoHS Annex IV) and to respond to the EU proposal to restrict the use of PFAS chemicals, resulting in a shift toward halogen-free laminates and fluorine-free surface treatments. Manufacturers are also beginning to track the embodied carbon in copper foil, creating another point of differentiation as OEMs expand supply-chain carbon accounting. In addition, VCP systems with closed-loop water recapture can have a smaller environmental footprint than traditional permanganate-based chemical desmear processes.
The creation of new RFQ line items creates opportunities for suppliers to operationalize ESG compliance through per-lot carbon-footprint reporting, an ISO 14064-1 greenhouse-gas inventory report, material-level REACH and RoHS 3.0 compliance certificates, and a PFAS phase-out roadmap with a defined completion window.
ESG compliance is not an increased cost, it is simply required to maintain access to the market for European medical device suppliers. An initial investment in building the quality gate infrastructure described in this guide will provide most of the necessary ESG compliance documentation as a byproduct of implementing the quality gates.
Your Medical PCB Procurement Action Plan
The production process to produce medical-grade printed circuit boards (PCBs) contains 42 steps consisting of 34 enabling steps supporting eight critical gates. Facilities that follow all of the critical gates defined by this process generally achieve stronger first-pass performance than facilities that do not follow these steps rigorously.
This document has defined how each of the critical gates correlates to the physical failure mechanism associated with the gate and the engineering parameters used to verify the gate and the procurement documentation requirements for each gate.
Three actions convert this framework into operational advantage.
First, for each medical PCB sourcing event, provide a stage-by-stage procurement table as an exhibit to the RFQ (request for quote). This table should include the production stages, engineering specifications, documentation requirements, and escalation triggers for that particular sourcing event, defined prior to sending out the first quote request.
Second, use the supplier selection framework to select the supplier based on the application scenario; for example, whether the application is implantable, high-frequency RF, or portable diagnostic, then match that scenario with the process-gate priorities, substrates, surface finishes, and certification requirements.
Third, ensure compliance with 2026 qualification expectations before the supplier is fully qualified. The new requirements regarding FDA SPDF cybersecurity documentation, EU CSRD carbon-footprint reporting, and IEC 81001-5-1 are changing how suppliers are qualified. Suppliers who have integrated HBOM (Hardware Bill of Materials) traceability — supporting the OEM’s SBOM obligations under FDA Section 524B —, ISO 14001-compliant environmental management, and DHR-linked RFID packaging are well-positioned; suppliers who are not fully compliant may experience qualification delays that can cascade into product-launch timelines.
Advanced Surface Finish Comparison for Medical-Grade PCB Assembly
| Surface Finish | Specification / Thickness | Shelf Life | IPC Standard | Medical Suitability | Reflow Cycles | Typical Medical Use |
|---|---|---|---|---|---|---|
| ENIG | Ni 3.0–6.0 μm / Au 0.05–0.10 μm | 12 months | IPC-4552A Rev. A | Excellent | 3–4 | Patient monitoring, defibrillator |
| Immersion Silver | Ag ≥0.20 μm | 6–12 months | IPC-4553A | Good | 2–3 | Disposable diagnostic cartridge |
| OSP | 0.20–0.50 μm organic film | 3–6 months | IPC-J-STD-003C | Poor | 1–2 | Non-critical consumer wellness |
| ENEPIG | Ni 3–6 μm / Pd 0.05–0.15 μm / Au 0.03–0.08 μm | 24+ months | IPC-4556A | Superior | 5+ | Implantable neurostimulators |
| Hard Electrolytic Au | Au ≥1.27 μm (50 μin) over Ni | Indefinite | MIL-G-45204 / ASTM B488 | Excellent | Unlimited | Surgical interface connectors |
| Immersion Tin | Sn 0.8–1.2 μm | 6 months | IPC-4554 | Fair | 2–3 | Press-fit backplane in imaging |
| HASL (Pb-free) | Sn/Ag/Cu 5–25 μm | 6–12 months | J-STD-006 | Poor | 2–3 | Legacy non-critical PSU boards |
ENEPIG is the finish most commonly used when black-pad risk avoidance, reflow-cycle endurance, and shelf-life stability are all high priorities, especially in implantable applications. For high-frequency RF designs, ENIG often remains the default option until assembly complexity or reflow demands justify an upgrade to ENEPIG. Within the process-gate framework defined in this document, surface-finish selection should be treated as a prerequisite for downstream verification of compliance. Together, the compliance verification tables, supplier selection framework, and documentation traceability architecture provide the documentation needed to support medical-quality audits.
Frequently Asked Questions
Q1. How long does a medical-grade 42-step PCB production cycle take from Gerber to shipment?
For boards with more than 8 layers, the cycle from Gerber submission to shipment commonly runs over multiple business weeks. Boards that utilize higher-layer HDI designs and sequential lamination require additional time for the lamination cycle. The major variables impacting turnaround time are the number of sequential lamination cycles and the turnaround for process-gate verifications, specifically TDR coupon testing and ionic cleanliness measurement. Expedited production programs can compress the schedule, but quality-gate verifications cannot be accelerated beyond what Class 3 compliance allows.
Q2. What drives the cost difference between a 12-layer and a 20-layer HDI medical board?
There are three principal contributors to the cost difference between a lower-layer and a higher-layer HDI medical board. First, the number of sequential lamination cycles increases significantly. Second, fine-line patterning yields are reduced because seed-layer uniformity becomes harder to hold across the greater number of imaging and plating cycles required to produce the final PCB. Third, material costs are higher because of the larger quantity of high-performance laminates, prepreg materials, and copper foil, with the premium increasing further when low-loss materials are included for signal-critical layers.
References & Sources
- IPC-6012 – Revision E – Addendum – Medical. Medical Applications Addendum to IPC-6012E Qualification and Performance Specification for Rigid Printed Boards — IPC
- ISO 13485:2016 – Medical devices — Quality management systems — Requirements for regulatory purposes — ISO
- IEC 81001-5-1:2021 — Health software and health IT systems safety, effectiveness and security – Part 5-1: Security – Activities in the product life cycle — IEC
- Quality Management System Regulation (QMSR) | FDA — U.S. Food and Drug Administration
- Cybersecurity in Medical Devices: Quality Management System Considerations and Content of Premarket Submissions | FDA — U.S. Food and Drug Administration
- Use of International Standard ISO 10993-1, “Biological evaluation of medical devices – Part 1: Evaluation and testing within a risk management process” Guidance for Industry and Food and Drug Administration Staff September 2023 | FDA — U.S. Food and Drug Administration
- eCFR :: 21 CFR 806.10 — Reports of corrections and removals. — Electronic Code of Federal Regulations
- Corporate sustainability reporting – Finance – European Commission — European Commission
- A Review of the Biocompatibility of Implantable Devices – PMC — PubMed Central

